Relapsing Remitting Multiple Sclerosis (RRMS)
Six European clinical neurologists met in December 2015 to finish and validate a model initially constructed by Merck KGaA staff of the benefit-safety balance of six RRMS drugs.
The clinicians considered seven favourable effects and 11 unfavourable effects, which are listed and defined in the Effects Table at the bottom of this page.
The clinicians considered seven favourable effects and 11 unfavourable effects, which are listed and defined in the Effects Table at the bottom of this page.
The options
The total benefit-safety balance is shown here on a 100-point scale. Green best; Red worst.
The total benefit-safety balance is shown here on a 100-point scale. Green best; Red worst.
- Dimethyl fumarate 63
- Cladribine 62
- Natalizumab 56
- Alemtuzumab 56
- Teriflunomide 49
- Fingolimod 48
The numbers represent the overall added clinical value of each drug. They take into account available data for the effects and judgements by the experts about the clinical relevance of the effects. Reductions in the frequency of MS symptoms define each drug's favourable effects, and frequencies of side effects are classed as unfavourable effects. The weighted sum of the two figures for each drug gives its overall benefit-safety balance.
The numbers represent the overall added clinical value of each drug. They take into account available data for the effects and judgements by the experts about the clinical relevance of the effects. Reductions in the frequency of MS symptoms define each drug's favourable effects, and frequencies of side effects are classed as unfavourable effects. The weighted sum of the two figures for each drug gives its overall benefit-safety balance.
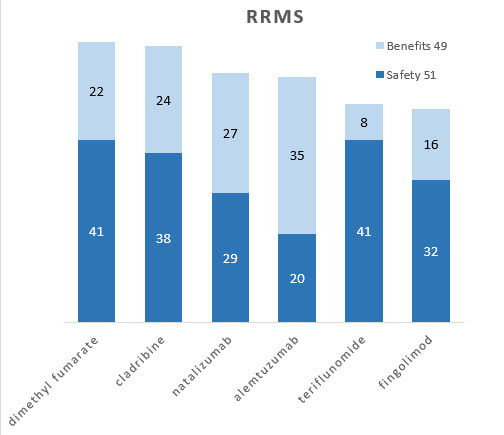
Bar graphs
Separate contributions of benefits and safety to the overall totals show different profiles. For example, dimethyl fumarate and cladribine are about equal overall, but the latter is more beneficial while the former is safer.
Separate contributions of benefits and safety to the overall totals show different profiles. For example, dimethyl fumarate and cladribine are about equal overall, but the latter is more beneficial while the former is safer.
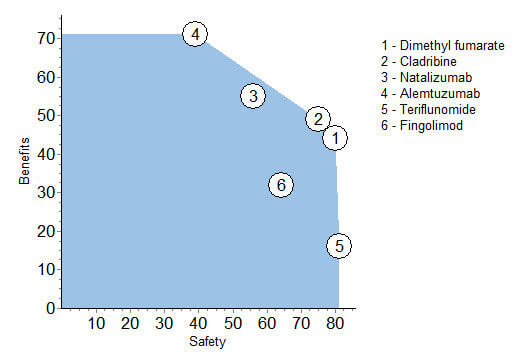
Benefits versus safety
Another way to look at the benefit and safety information is to show each drug's two figures before the 64-36 weights are applied.
Another way to look at the benefit and safety information is to show each drug's two figures before the 64-36 weights are applied.
This figure facilitates comparisons of the drugs. Note that both dimethyl fumarate and cladribine are better at this level of analysis for safety and benefits than fingolimod, and their benefits outweigh teriflunomide's slight advantage in safety. It is clear that 1, 2, 3 and 4 are nearly on a straight line; more benefit means less safety.
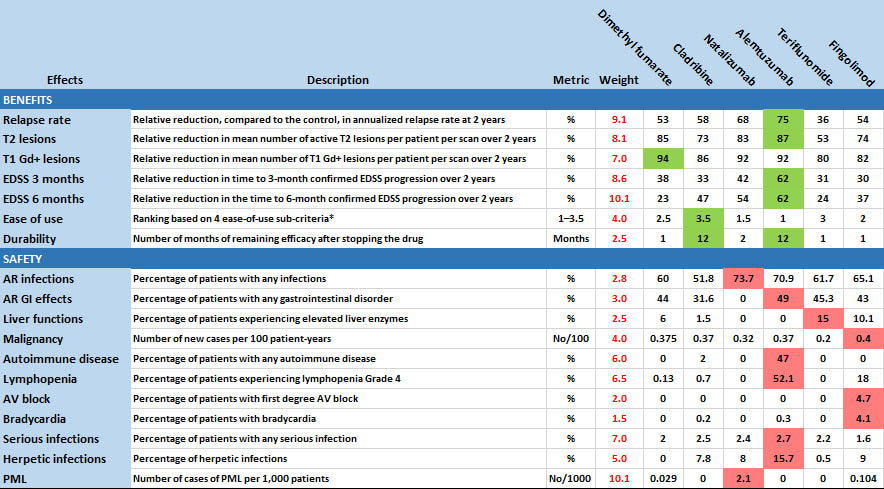
Effects Table
All the above is based on the following data, culled from available public sources, and experts' weight assessments about the relative clinical relevance of the effects. To support patient-oriented prescribing based on the evidence, green shading identifies the best drug for each favourable effect while red shows the worst drug for each unfavourable effect.
All the above is based on the following data, culled from available public sources, and experts' weight assessments about the relative clinical relevance of the effects. To support patient-oriented prescribing based on the evidence, green shading identifies the best drug for each favourable effect while red shows the worst drug for each unfavourable effect.
* (1) oral vs iv, (2) few or many doses, (3) monitoring during administration (Y or N) and (4) Co-administration of other drugs (Y or N). Bold Face means preferred.
AR=adverse reaction; AV=atrioventricular; EDSS=expanded disability status scale; Gd+=gadolinium enhanced; PML=progressive multifocal leukoencephalopathy.
AR=adverse reaction; AV=atrioventricular; EDSS=expanded disability status scale; Gd+=gadolinium enhanced; PML=progressive multifocal leukoencephalopathy.
Note that the weighted scores are relative and zero does not mean no value, so only differences between numbers, not their ratios, make sense for comparing the clinical value they represent.
Reference: Vermersch, P., Martinelli, V., Pfleger, C., Rieckmann, P., Alonso-Magdalena, L., Galazka, A., . . . Phillips, L. (2019). Benefit-risk Assessment of Cladribine using Multi-Criteria Decision Analysis (MCDA) for Patients with Relapsing-Remitting Multiple Sclerosis. Clinical Therapeutics, 41(2), 249-260.